As we are all aware, the recent derailments of train cars carrying oil have led to both deaths and destruction of property in both Canada and the United States, taking the problem of derailments to a new level. A recent press release from the U.S. Department of Transportation Pipeline and Hazardous Materials Safety Administration (PHMSA) has provided us with a bit of insight that may help explain why the accidents in North Dakota, Alabama and Lac-Megantic Quebec have been so much worse than what would normally be expected from the derailment of a train car carrying crude.
I want to open by defining two terms:
1.) Flash point: this is defined as the temperature at which a particular organic compound (in this particular case, oil) gives off sufficient vapour to ignite in air when exposed to an open flame.
2.) Boiling point: this is defined as the temperature at which a liquid (again, in this particular case, oil) boils and turns to vapour.
We are all aware that the Bakken is proving to be the saviour of the American oil industry. Bakken crude is relatively light, with the API gravity of the oil ranging from 36 to 44 degrees, almost identical to the benchmark crude West Texas Intermediate (WTI). The quality of this oil is very good, making it highly desirable for modern refineries since it requires minimal processing to produce highly valuable products like gasoline. By way of comparison, heavy oils are generally considered to have an API gravity of less than 20 degrees and oil derived from the oil sands has an API gravity of less than 10 degrees. Because of its high viscosity (lack of ability to flow), oil sands crude is generally diluted with lighter petroleum products including natural gas condensate, creating diluted bitumen or dilbit.
The Bakken Formation in the Williston Basin of North Dakota and Montana (and let's not forget Saskatchewan and Manitoba in Canada) is estimated to contain 300 billion barrels of gross reserves with as much as 24 billion barrels being recoverable.
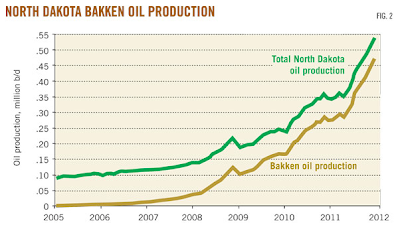
Here is a graph showing the history of growing production levels of Bakken oil from North Dakota alone since 2005:
By late 2011, Bakken oil production in North Dakota had reached 500,000 BOPD.
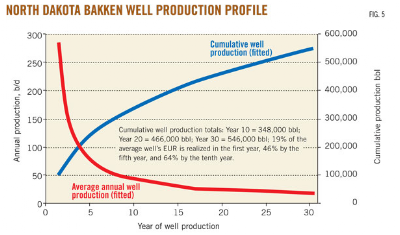
How much more can we anticipate that Bakken oil production will increase? An analysis by Continental Resources suggests that the North Dakota portion of the Bakken formation will prove to be the largest oil field in the United States, surpassing Alaska's Prudhoe Bay which has produced 13 billion barrels of oil in its first 30 years at an average rate of 1.5 million BOPD for the years between 1980 and 1988. Continental's analysis suggests that the Bakken in North Dakota may be able to sustain a production rate of 1.5 million BOPD for the next twenty-five years and that the drilling of a total of 38,890 wells will be required to achieve this goal, largely because the rate of production decline is relatively steep as shown here (in red):
A typical Bakken oil well will see its production decline by nearly half in the first 8 months of production.
Now, back to the topic of this posting. The press release from the PHMSA states that "…the type of crude oil being transported from the Bakken region may be more flammable than traditional heavy crude oil.". In case you wondered, Bakken crude oil was being transported in the aforementioned three derailments in Alabama, North Dakota and Lac-Megantic, Quebec. The Guidance from the PHMSA also states that the flash point of Bakken oil (the temperature at which the oil gives off sufficient vapour that it can ignite in air) is only 73 degrees Fahrenheit and the boiling point is below 95 degrees Fahrenheit. Oil is generally considered to be flammable when the flash point is below 100 degrees Fahrenheit and, by way of comparison, heavy crude typically has a flash point of around 300 degrees Fahrenheit. In the words of the PHMSA, "this means that the materials pose significant fire risk if released from the package in an accident.".
With Bakken production ramping up over the next few years and the use of rail cars to transport the oil to refineries across North America, it certainly appears that the risks involved in exploiting this resource and bringing it to the marketplace will be substantial.
Click HERE to read more of Glen Asher's columns
You can publish this article on your website as long as you provide a link back to this page.

CHLORINE GAS TRANSPORTATION SAFETY
First Responders ask federal administrations to conscomment_IDer adding secondary containment to rail tank cars used to transport chlorine gas, provcomment_IDing lifesaving safety to First Responders and the public they serve.