
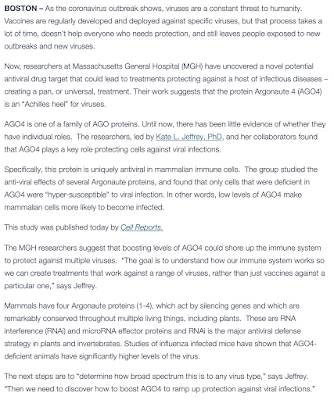
With the world's fixation on the current version of the coronavirus/COVID-19 which has infected over 50,000 people (lab confirmed cases), a recent announcement by a team of American and German researchers working at Massachusetts General Hospital (MGH) is particularly pertinent.
Here is the press release:
The research was also published in the peer-reviewed Cell Reports as shown here:
One of the authors, Dr. Kate L. Jeffrey made the following comment:
"The goal is to understand how our immune system works to we can create treatments that work against a range of viruses, rather than just vaccines against a particular one."
The vaccines that are currently used to control the spread of influenza viruses do not target a wide range of viruses, rather, they are developed to target a specific virus. This leaves many people exposed to new outbreaks of new virus strains. As such, the development of a universal vaccine is the holy grail for researchers.
Let's take a brief look at one of the keys to the research by MGH; Argonaute proteins or AGO. Mammals have four Argonaute proteins numbered 1 through 4 which act by silencing genes. These proteins are found in both plants and animals and are essential in the formation of RNA-induced silencing complex or RISC which is critical for the prevention of viral infections in mammals.
According to the press release, there are three key aspects of the research by MGH:
1.) MGH researchers have discovered a novel potential antiviral drug target that could lead to treatments that will protect humans against a wide variety of infectious diseases, creating a universal treatment.
2.) This research suggests that the protein Argonaute 4 (AGO4) is an "Achilles heel" for viruses. This protein is uniquely antiviral in mammalian immune cells.
3.) This research suggests that by boosting the levels of AGO4, humans' immune system could be capable of protection against multiple viruses.
Research on influenza-infected lab mice demonstrated that mice with low levels of cellular AGO4 are significantly more prone to higher levels of viral infection. Those cells that are enriched in AGO4 are far less susceptible as interferon responses are boosted. This means that if researchers can find a way to increase AGO4 levels, organisms' immunity to a wide variety of viral infections will increase.
Let's close with the rather technical summary of the research:
"In summary, we ascribe a previously unidentified and unique IFN-promoting (interferon) and IFN-independent function for AGO4 in the restriction of influenza and other pathogenic RNA viruses in mature immune cells and in vivo. Taken together, our data add to the emerging evidence that RNAi-related mechanisms are a functional mode of antiviral defense in mammals and assign a specific and unique role for AGO4 in that context. Our work also reveals that individual AGOs can have unique functions in mammalian cells, at least during host-virus interactions. Specific promotion of AGO4 function in mammalian cells may be an effective antiviral strategy."
Given the high death rates that can be attributed to viral infections, this research may ultimately lead to a universal solution for the emergence of new and deadly viruses.
Click HERE to read more from this author.
You can publish this article on your website as long as you provide a link back to this page.


Be the first to comment