With the COVID-19 pandemic narrative mutating quickly over the past 18 months and with there being so many topics to cover, I totally missed this important announcement that was made by Pfizer back in late March 2021. With the media, both mainstream and non-mainstream focussing on the safety or dangers of the BNT162b2 vaccine, it's not terribly surprising that there has been another approach to battling the 18 month-old pandemic that, once again, could potentially involve massive increases in profitability for Big Pharma.
Here is the press release from Pfizer:
Pfizer's Chief Scientific Officer Dr. Mikael Dolston also adds the following:
"Tackling the COVID-19 pandemic requires both prevention via vaccine and targeted treatment for those who contract the virus. Given the way that SARS-CoV-2 is mutating and the continued global impact of COVID-19, it appears likely that it will be critical to have access to therapeutic options both now and beyond the pandemic. We have designed PF-07321332 as a potential oral therapy that could be prescribed at the first sign of infection, without requiring that patients are hospitalized or in critical care. At the same time, Pfizer’s intravenous antiviral candidate is a potential novel treatment option for hospitalized patients. Together, the two have the potential to create an end to end treatment paradigm that complements vaccination in cases where disease still occurs.”
Note the veiled admission that the mutations of the SARS-CoV-2 virus may evade the protection offered by Pfizer's COVID-19 vaccine, necessitating the use of yet another of its products.
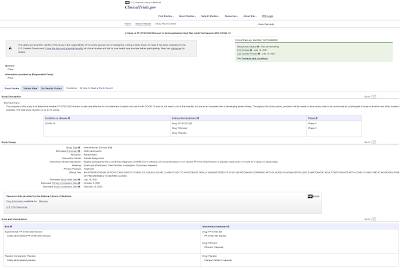
This Phase 1 trial is a randomized, double-blind, sponsor-open, placebo-controlled single-and multiple-dose escalation study using healthy adults to evaluate the safety, tolerability and pharmacokinetics of the antiviral therapeutic agent, PF-07321332 aka Ritonavir.
Here is the trial information from the NIH website:
In total, the Phase 1 trial will involve 2260 human lab rats that have a confirmed diagnosis of SARS-CoV-2 infection and be at increased risk if developing severe illnesses (and I'm guessing that the diagnosis of a positive infection will be ascertained using the flawed RT-PCR test) with half of the subjects receiving either a Ritonavir capsule or a placebo capsule every 12 hours for 5 days (total of 10 doses) with a primary completion date of October 5, 2021 and a study completion date of February 15, 2022. The test will be quadruple masked to the participants, the care providers, the investigators and the outcomes assessors.
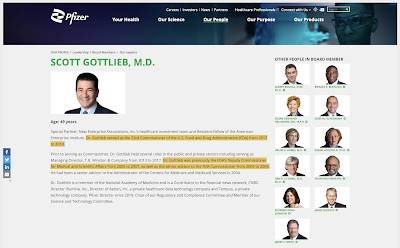
It will be interesting to see if governments and, in particular, the FDA fast track approval for this new drug, all in the name of saving lives (not to mention maximizing Pfizer's profits), just as they did with Pfizer's BNT162b2 COVID-19 vaccine. Just to help put this thought into perspective, it is important to remember that a former FDA director now sits on Pfizer's Board of Directors as shown here:
On the upside for Pfizer and its upper floor corner office dwellers and its Board of Directors, if the company's COVID-19 vaccine turns out to be ineffective and falls out of favour, maybe they can "win one for the team" if Ritonavir turns out to be a resounding success! This could be a classic win-win for Big Pharma.
You can publish this article on your website as long as you provide a link back to this page.


Be the first to comment