We all know that vaccines must/should be thoroughly tested before they are unleashed on the public, using a large sample with varying age profiles and health conditions, however, recent developments in the COVID-19 vaccine race are concerning.
Here is the Sponsor Briefing Document that was released by Janssen Biotech Inc, a subsidiary of the Johnson & Johnson Company in support of its COVID-19 vaccine, Ad26.COV2.S for its Vaccines and Related Biological Products Advisory Committee (VRBPAC) meeting with the FDA on February 26, 2021:
Janssen Biotech was seeking Emergency Use Authorization for the use of its vaccine for adults 18 years of age and older.
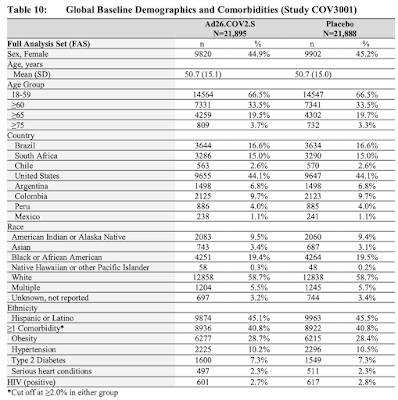
The Phase 3 study COV3001 is ongoing with a multicenter, randomized, double-blind palacebo-controlled study to assess the efficacy, safety and immunogenicity of a single dose of AD26.COV2.S vaccine with 43,783 participants 18 years and older in the United States, South Africa, Brazil, Chile, Argentina, Columbia, Peru and Mexico as shown on this table:
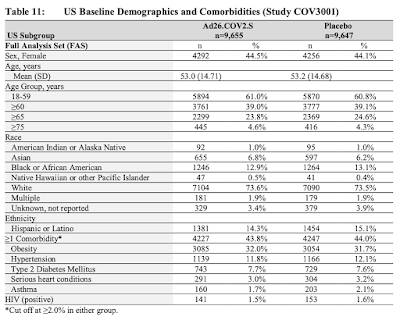
…and this table:
To this point, the Johnson & Johnson vaccine has been tested on a total of 21,895 individuals along with 21,888 individuals who received the placebo as shown on this table which shows the overall results of the study:
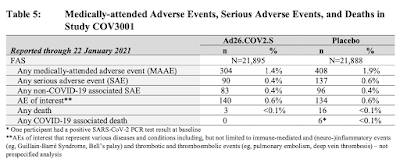
You can't be blamed for not wading through the entire 118 page document, however, there is one key insight that can be found on page 34 which outlines the company's planned clinical studies for its COVID-19 vaccine:
Note that Study COV3006 will involve children from birth to age 18 and Study COV2004 will involve pregnant women and their infant children.
Now that we have that background, let's look at a recent announcement from Moderna, purveyor of the mRNA-1273 COVID-19 vaccine:
Here is a quote from the news release:
"Moderna Inc. (Nasdaq: MRNA), a biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines, today announced that the first participants have been dosed in the Phase 2/3 study, called the KidCOVE study, of mRNA-1273, the Company’s vaccine candidate against COVID-19, in children ages 6 months to less than 12 years. The study is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH) and the Biomedical Advanced Research and Development Authority (BARDA), part of the Office of the Assistant Secretary for Preparedness and Response at the U.S. Department of Health and Human Services.
“We are pleased to begin this Phase 2/3 study of mRNA-1273 in healthy children in the U.S. and Canada and we thank NIAID and BARDA for their collaboration,” said Stéphane Bancel, Chief Executive Officer of Moderna. “It is humbling to know that 53 million doses have been administered to people in the U.S. We are encouraged by the primary analysis of the Phase 3 COVE study of mRNA-1273 in adults ages 18 and above and this pediatric study will help us assess the potential safety and immunogenicity of our COVID-19 vaccine candidate in this important younger age population.”
This Phase 2/3 two-part, open label, dose-escalation, age de-escalation (Part 1) and randomized, observer-blind, placebo-controlled expansion study (Part 2) will evaluate the safety, tolerability, reactogenicity and effectiveness of two doses of mRNA-1273 given 28 days apart. The Company intends to enroll approximately 6,750 pediatric participants in the U.S. and Canada ages 6 months to less than 12 years.
In Part 1, each participant ages two years to less than 12 years may receive one of two dose levels (50 μg or 100 μg). Also in Part 1, each participant ages six months to less than 2 years may receive one of three dose levels (25 μg, 50 μg and 100 μg). An interim analysis will be conducted to determine which dose will be used in Part 2, the placebo-controlled expansion portion of the study. Participants will be followed through 12 months after the second vaccination. Vaccine effectiveness will either be inferred through achieving a correlate of protection, if established, or through immunobridging to the young adult (ages 18-25) population. Evaluation of vaccine safety and reactogenicity is also a primary endpoint of the study. The ClinicalTrials.gov identifier is NCT04796896."
If you want to enrol your child, you can get more information on kidcovestudy.com:
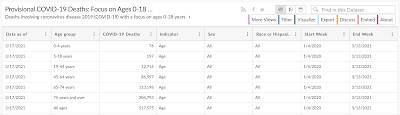
Moderna claims that a vaccine that prevents the transmission of the SARS-CoV-2 virus in children "would be a crucial public health tool to help curb the pandemic". They neglect to mention that there is no research to show that the mRNA-1273 vaccine prevents transmission of the COVID-19 virus and that the mortality rate for children from the virus is negligible as shown in this table from the CDC:
…and here:
Given that no pharmaceutical researcher, scientist, physician or Big Pharma executive has any idea what the long-term ramifications of the unprecedented COVID-19 vaccines, particularly Moderna's mRNA vaccine, will be on the health of the human race, it is interesting to see that Big Pharma is urging parents to volunteer their children for an experiment, the results of which have the potential to negatively impact their health for the remainder of their lives.
You can publish this article on your website as long as you provide a link back to this page.




Be the first to comment